Example of electron capture8/18/2023 Whereas beta decay can occur spontaneously when energetically allowed, for an electron capture the weak forces require that the electron comes into close contact with a proton of the nucleus. 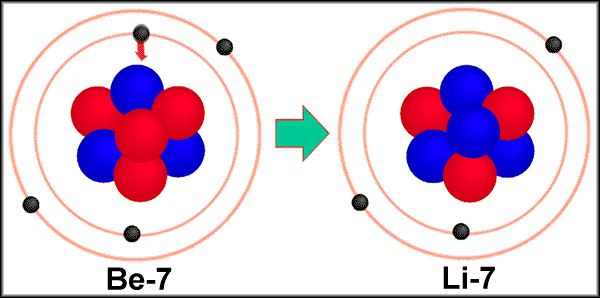
Electron capture occurs much less frequently than the emission of a positron. Weak forces are behind positron emission and electron capture. This explains why electron capture is difficult and therefore rare.Įlectron capture : a difficult and rare process Even the innermost K-layer electrons are far from the very small volume of the nucleus where the weak forces responsible for the capture operate and transform the electron into a neutrino. Most of the electrons orbit the nucleus at distances large compared to the nucleus. The captured electron belongs to the group of electrons orbiting around the nucleus. Ordinary beta-minus decay has no competitor on Earth however to reduce an excess of neutrons, since the capture of positrons would occur in an world made of antimatter. Electron capture, along with beta-positive decay, is Nature’s way of guaranteeing that no nucleus becomes too proton-heavy. The capture of an electron has the same effect on a nucleus as the emission of a positron: one of its protons transforms into a neutron, diminishing the global electric charge of the nucleus by 1 unit. The electron capture trigger the emission of an invisible neutrino by the nucleus. The best-known example is of potassium 40 : 11% of the nuclei of that isotope of potassium present in our body decay by electronic capture. In both cases, practically all the enegy released is carried by light particles.Įlectron capture is a comparatively minor decay mode caused by the weak force. There is no such energy threshold in the case of electron capture (bottom). The emission of a positron and the capture of an electron are twin reactions both resulting in the diminution of the number of protons by 1 (from Z to Z-1) and the production of a neutrino.The positron observed in the final stage of the beta decay (top) is a new particle requiring the 0.511 MeV of its rest mass energy to be created. The electron is not a reactant but a product.Positron emission versus electron capture So this would be an example of electron capture, which is the opposite of beta decay or a mission invaded a care admission. We absorb an electron, so 207 plus zero is 207 80 to minus one is 81. 
And finally we have led to 07 so this would be 207 The atomic number of lead is 82. So there's a lot of elements that are named after people we might have heard off or may not have heard off. You'll find that a lot of elements around Nobel, you are named after states named after countries named after just inventors and physicists and scientists. So to 60 plus zero is to 60 one or two minus one is one on one. So here, we're gonna have no belly, um, to 60 which is We absorbing electron. Give me 103 And that will be L R for the next one. So to 63 plus zero gives me to 63 104 minus one. So here, Rutherford has an atomic number of 104 here. So remember Rutherford was the guy who came up with the three major types of nuclear reactions, and they named Element 104 after him. We have to say right balance nuclear equations for each of the following elements after undergoing electron capture. Instead of doing beta decay, remission were doing beta capture, a k A. So that represents the opposite of beta decay. 23 and then 87 minus one is gonna give me 86. What effect is that gonna do? Well, it's gonna be 2. So here capture means that this electron is not gonna be a product, but it's gonna be a reacted. And here we'll say we're dealing with isotope to 23. So let's think of an example we could deal with Francie um, which is the metal most to the left and the lowest down group one A. So here we're gonna say electron capture involves the absorption, often electron, which remember, we saw as this symbol find unstable nucleus and is represented by the following reaction. The electron will be a product, but invade a capture or electron capture. They're both dealing with an electron beta decay. 
So when we say electron capture, we're really saying beta capture. I remember an electron is really just a beta particle. And here we're talking about an electron. A mission means that you're particle will be a product, but capture means that it'll be a reacted. Now, the word capture we've been talking about this there is decay in a mission versus capture. Hey, guys, In this new video, we're gonna take a look at electron capture.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
